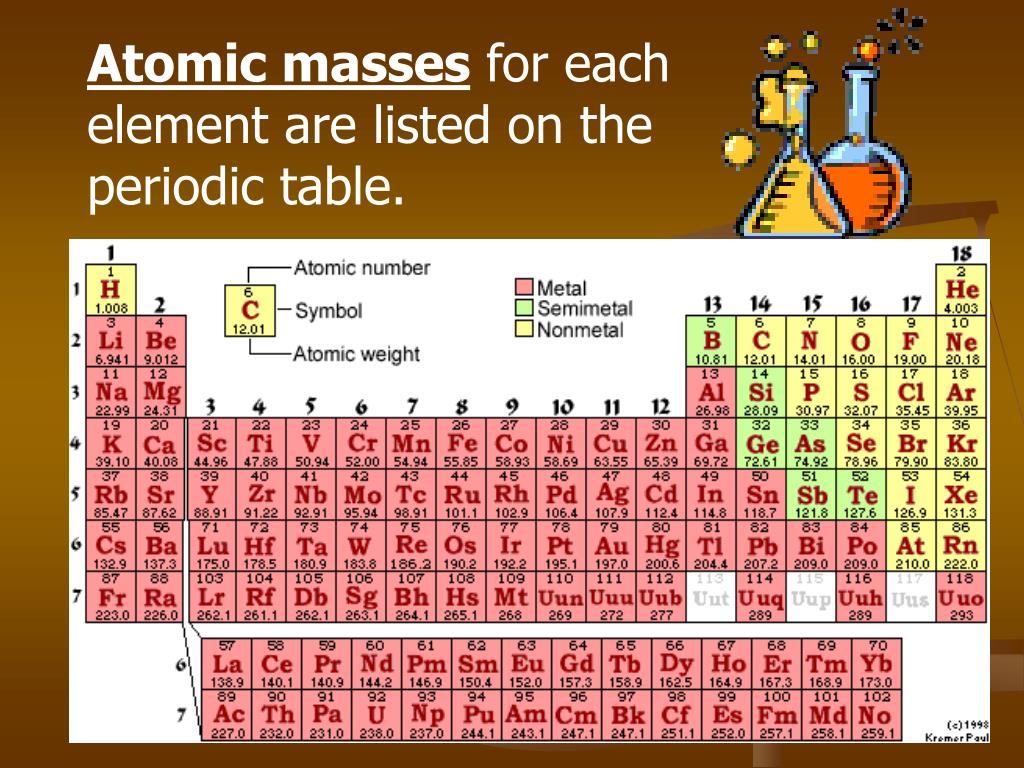
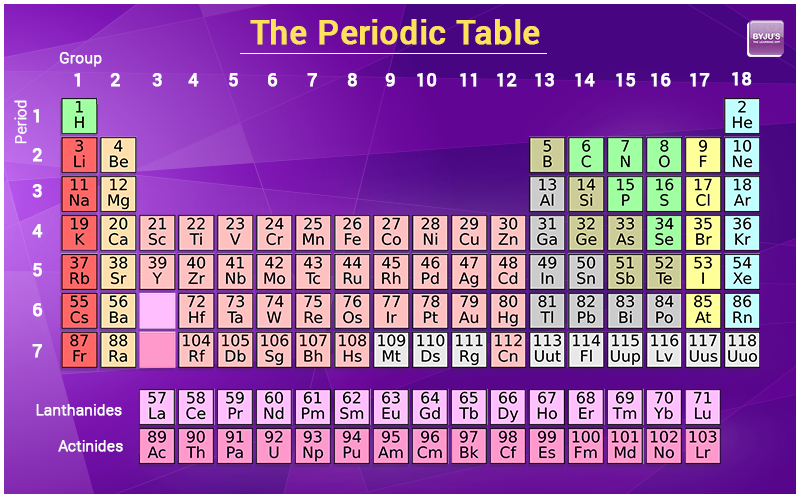
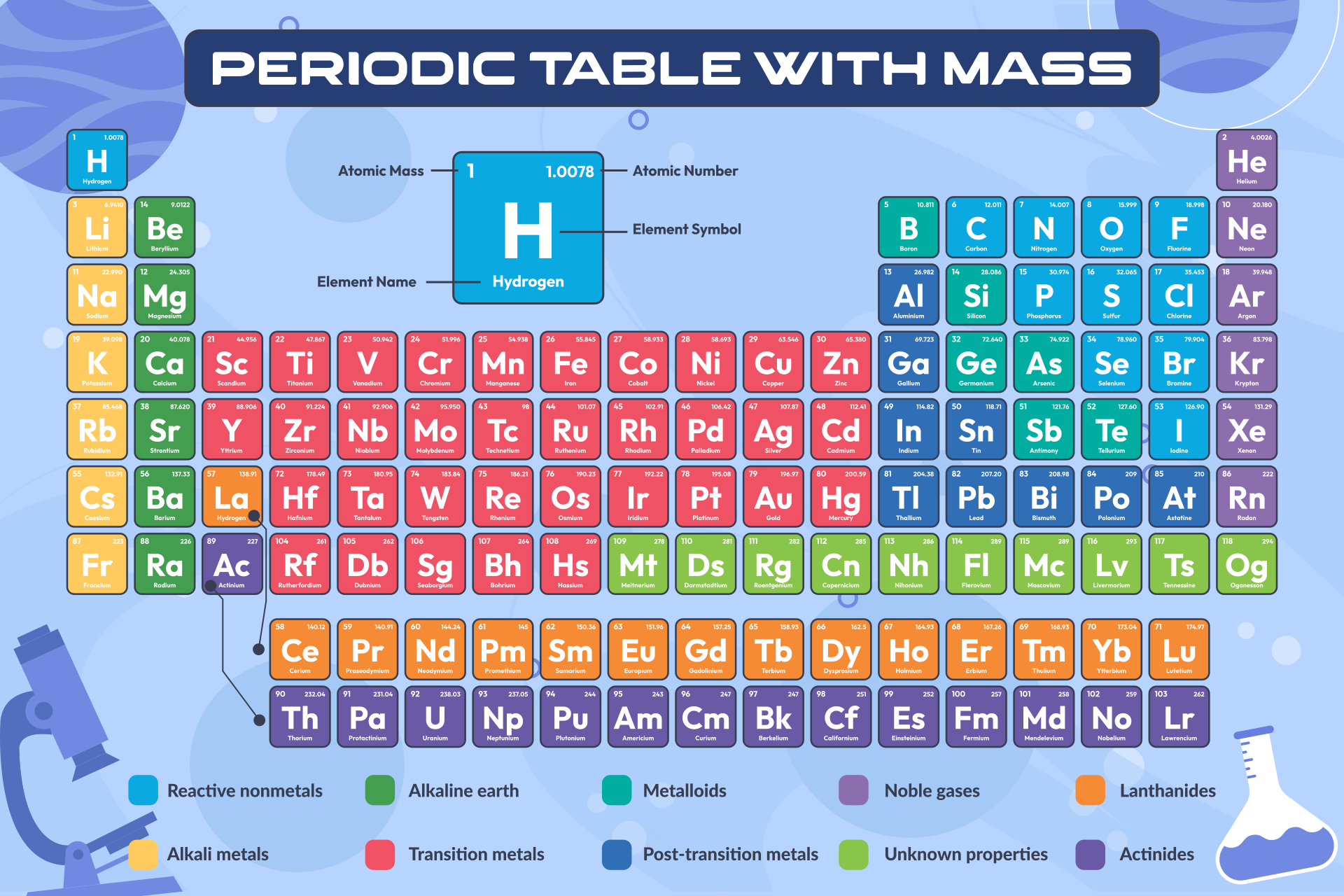
One atomic mass unit is defined as 1/12th of the mass of a carbon-12 atom, which is approximately 1.66 x 10-27 kg.Ītomic mass is typically calculated by taking into account the natural abundance of isotopes of an element and their individual masses. The atomic mass unit (amu) is the standard unit used to express atomic mass. The atomic mass is the sum of the masses of its protons, neutrons, and electrons an electron’s mass is so small in comparison to the other particles that its mass is simply that of the nucleus (protons and neutrons).The relative abundance of isotopes in a naturally occurring element is used to calculate the average mass of an element. /periodic-table-of-the-elements-2017--illustration-769723031-5aa02f9b04d1cf00386ccf7c.jpg)
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
